Mechanical cues that shape the biology of fibroblasts in Lupus Nephritis
Lead: Ayesha Budhwani
Team Members: Dr. Crosslee Titus, Vinaika Maruvada, Wasim Shaik, Ali Sherwani, Lana Nguyen
Project Summary:
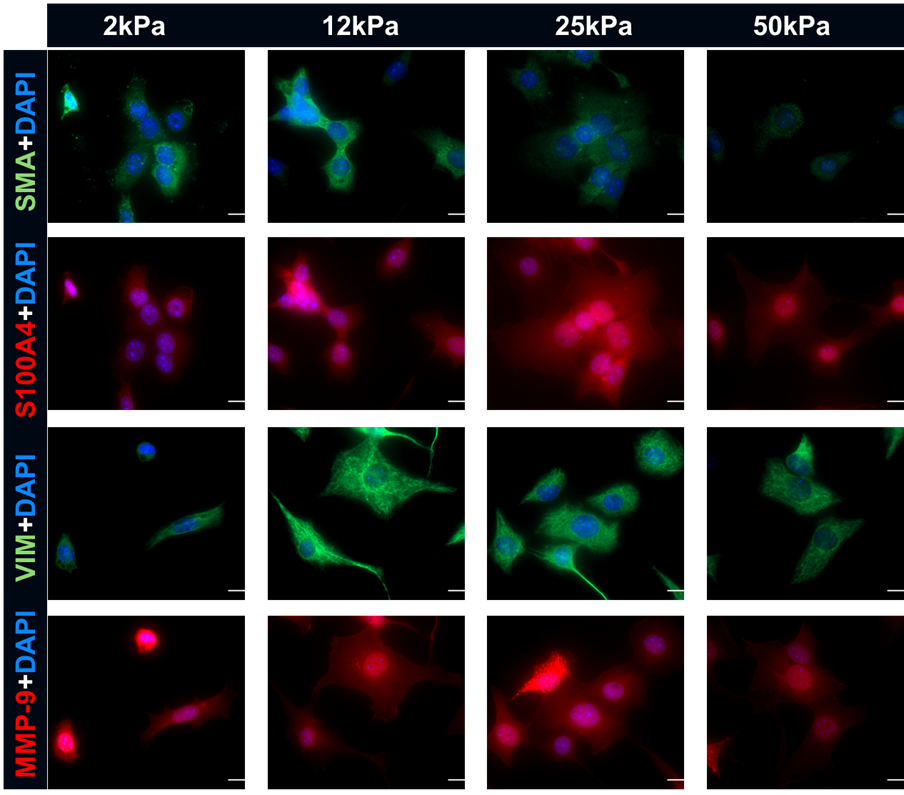
This project seeks to identify the specific mechanical and biochemical signals within the renal microenvironment that drive the progression of Lupus Nephritis. While much research focuses on systemic immune triggers, this study investigates how the local environment, specifically tissue stiffness, LN and differentially expressed extracellular matrix proteins interact to reprogram renal fibroblasts and tubular epithelial cells. By using tunable 3D hydrogels and cell culture models, we evaluate how these factors promote a transition towards a persistent, pro-fibrotic phenotype that leads to irreversible kidney damage.
What is already known in the field?
- It is well-established that inflammatory cytokines activate fibroblasts into myofibroblasts.
- Traditionally, ECM was viewed as a passive scar that resulted from damage, rather than an active agent that influences cell behavior in a paracrine fashion.
What is new?
- We are evaluating how the physical “hardening” of the kidney acts as a signal that cells sense and respond to via mechano-transduction.
- We are identifying how specific proteins increased in LN act as active ligands that shape cell biology rather than just providing structural support.
Why is this important?
Targeting the way cells “feel” their environment could provide a way to halt fibrosis without suppressing the immune system.
Ongoing/future steps:
- Establish in vitro models using stiffness-controlled hydrogels and LN-specific ECM proteins to map the thresholds of fibroblast and tubular cell activation.
- Conduct bulk-RNA sequencing to characterize the transcriptomic landscape and identify key regulatory pathways driving renal modeling.
- Translational studies in human lupus nephritis.