AI-Aided Assessment of Renal Pathology in Lupus Nephritis
Lead: Mohammad Daouk
Team Members: Dr. Anto Sam Crosslee Louis Sam Titus, Munim Ahmed, Surya Ummadi, Thanoj Aila
Collaborators: Dr. Hien Van Nguyen
Pathology Partners: Dr. Jan Becker (University Hospital of Cologne (Germany)), Dr. Anthony Chang (University of Chicago (IL, USA)), Dr. Neeraja Kambham (Stanford University (CA, USA)), Dr. Surya Seshan (Cornell University (NY, USA)), Dr. Cai Qi (UT Southwestern (TX, USA)), Dr. Luan Truong (Houston Methodist (TX, USA)), University of Bari (Italy)
Project Summary:
Lupus nephritis (LN) is a severe manifestation of systemic lupus erythematosus and remains a major cause of chronic kidney disease and kidney failure. Diagnosis and grading rely on renal biopsy interpretation, but real-world pathology workflows face substantial variability across institutions due to differences in stains, scanners, and tissue preparation protocols.
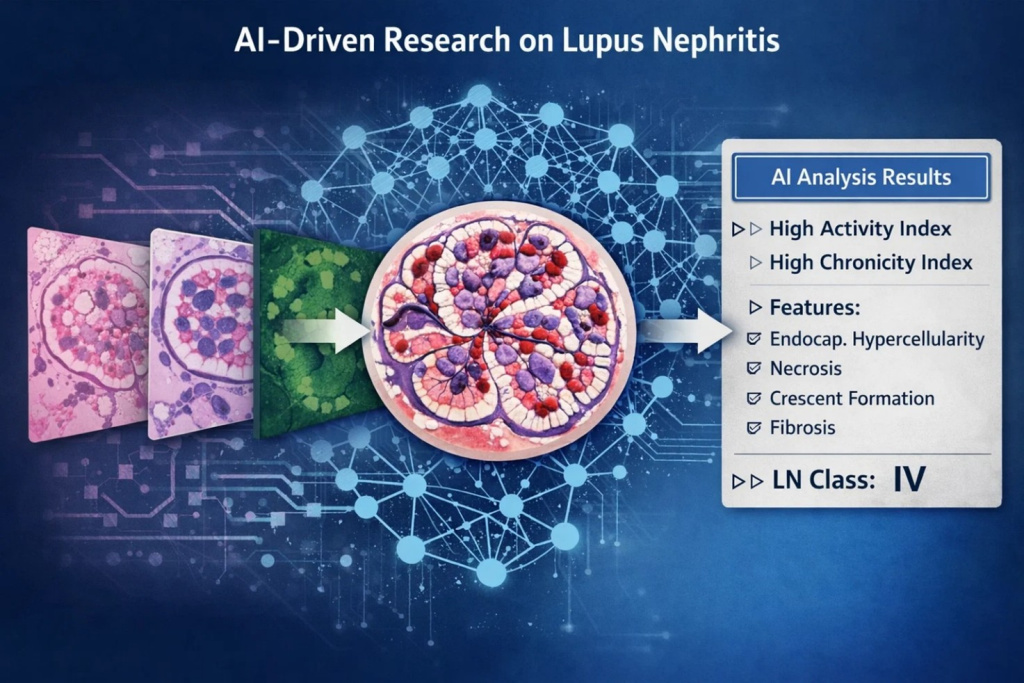
In this project, we are developing robust, uncertainty-aware AI models for LN pathology using renal biopsy whole-slide images (WSIs). Our pipeline is designed to (i) identify and segment key renal compartments (e.g., glomeruli and tubules), (ii) predict glomerular and tubular Activity Index (AI) and Chronicity Index (CI) components, and (iii) infer biopsy-level LN class alongside biopsy-level AI/CI scores. A central design principle is reliability under real-world variability, so the system explicitly detects, quantifies, and mitigates bias and domain shift across centers, stains, and processing pipelines.
What is already known in the field?
AI models can achieve strong performance in renal pathology tasks under controlled settings, but generalization often degrades when deployed across different centers, stains, scanners, and tissue preparation protocols. Uncertainty estimation and domain shift detection are increasingly recognized as critical for safe clinical translation.
What is new?
This work builds an end-to-end LN pathology pipeline that is explicitly uncertainty-aware and domain-robust. We have developed Bayesian deep neural network models that classify proliferative versus non-proliferative glomerular lesions while producing predictive uncertainty estimates. These uncertainty signals are used to flag out-of-distribution samples (e.g., unseen stains or center-specific shifts), support triage for expert review, and enable safer biopsy-level inference.
Why is this important?
LN pathology decisions directly impact treatment intensity and long-term outcomes, so unreliable model behavior under distribution shift is unacceptable. By integrating uncertainty quantification, bias detection/mitigation, rigorous cross-center evaluation, and continuous monitoring for drift, this project prioritizes safety, interpretability, and real-world deployability rather than single-site accuracy alone.
Ongoing/Future Steps
Ongoing work focuses on cohort expansion and curation of a large multi-institutional WSI dataset spanning PAS, H&E, Trichrome, and Jones stains, with multiple section levels per case to capture histologic heterogeneity. We are also developing segmentation foundation models to partition WSIs into core renal structures (including glomeruli and tubules), followed by structure-level AI/CI scoring models with uncertainty-aware aggregation. Finally, we will output biopsy-level LN class and calibrated AI/CI scores, and extend to a multimodal framework integrating light microscopy with immunofluorescence (IF), electron microscopy (EM), and clinical reports/metadata and treatment outcomes to better reflect real nephropathology workflows.