Targeting pro-fibrotic S100A4 in Lupus Nephritis
Leads: Crosslee Titus, Geethangili Madamanchi, Jayaraman Tharmalingam
Team Members: Kamala Vanarsa, Aalekhya Biswas
Collaborators: Calluna Pharma
Project Summary:
Renal fibrosis is the strongest predictor of long-term kidney failure in LN, yet effective anti-fibrotic therapies are lacking. This project focuses on S100A4, a pro-fibrotic and pro-inflammatory mediator, to define its spatial expression, cellular sources, upstream triggers, and therapeutic potential in LN.
What is already known in the field?
S100A4 (fibroblast-specific protein-1) is implicated in fibrosis across multiple organs and amplifies TGF-β, NF-κB, and epithelial–mesenchymal transition pathways. Elevated S100A4 has been reported in chronic kidney disease, but its cell-specific spatial distribution and functional relevance in LN are unknown.
What is new?
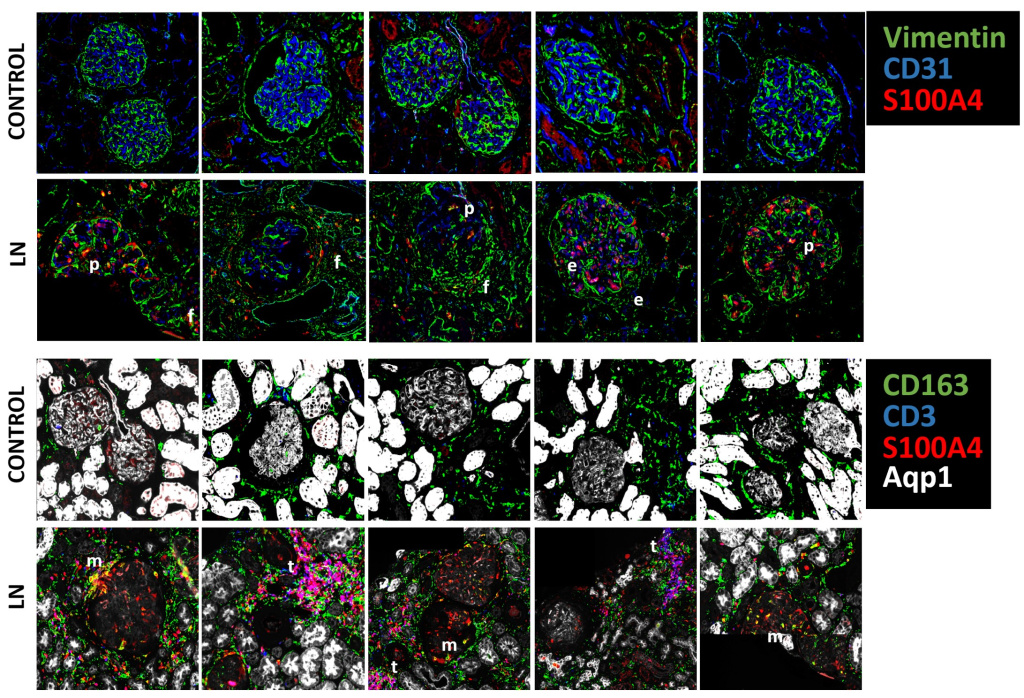
Using integrated spatial proteomics and single-cell spatial transcriptomics, we demonstrate that S100A4 is enriched in LN glomeruli and tubulointerstitial regions, with prominent expression in macrophages, T cells, fibroblasts, podocytes, and PECs. Spatial transcriptomics reveals strong correlations between S100A4 expression and pro-fibrotic, inflammatory, and hypoxia-associated pathways. In parallel, pharmacologic blockade and genetic ablation of S100A4 are underway in lupus-prone mouse models.
Why is this important?
These findings position S100A4 as a central, druggable mediator linking immune activation to fibrosis in LN. Targeting S100A4 offers a strategy to disrupt fibrosis while preserving immune function.
Ongoing/Future Steps
Complete pharmacologic / genetic S100A4 blockade studies in LN mouse models
Quantify fibrosis reduction and molecular signature upon S100A4 inhibition